 AUTHORS: Meisheng Ma,¹ Mihaela Stoyanova,² Griffin Rademacher,³ Susan K. Dutcher,² Alan Brown,³ and Rui Zhang,¹. CELL PRESS, Article | Volume 179, Issue 4, P909-922.E12, October 31, 2019.
AUTHORS: Meisheng Ma,¹ Mihaela Stoyanova,² Griffin Rademacher,³ Susan K. Dutcher,² Alan Brown,³ and Rui Zhang,¹. CELL PRESS, Article | Volume 179, Issue 4, P909-922.E12, October 31, 2019.
¹Department of Biochemistry and Molecular Biophysics, Washington University in St. Louis, School of Medicine, St. Louis, MO, USA
²Department of Genetics, Washington University in St. Louis, MO, USA
³Department of Biological Chemistry and Molecular Pharmacology, Blavatnik Institute, Harvard Medical School, Boston, MA, USA
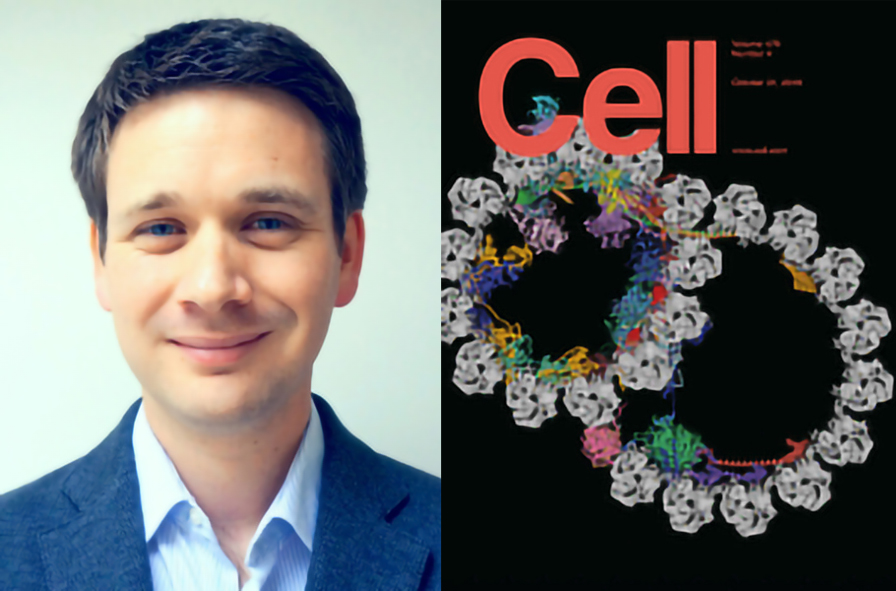
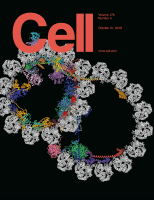
IN BRIEF: Visualizing axonemal microtubules and the proteins that decorate them, on the outside and inside, points to how the underlying periodic architecture supports cilia function.
SUMMARY: The axoneme of motile cilia is the largest macromolecular machine of eukaryotic cells. In humans, impaired axoneme function causes a range of ciliopathies. Axoneme assembly, structure, and motility require a radially arranged set of doublet microtubules, each decorated in repeating patterns with non-tubulin components. Single-particle cryo-electron microscopy is used to visualize and build an atomic model of the repeating structure of a native axonemal doublet microtubule, which reveals the identities, positions, repeat lengths, and interactions of 38 associated proteins, including 33 microtubule inner proteins (MIPs). The structure demonstrates how these proteins establish the unique architecture of doublet microtubules, maintain coherent periodicities along the axoneme, and stabilize the microtubules against the repeated mechanical stress induced by ciliary motility. This work elucidates the architectural principles that underpin the assembly of this large, repetitive eukaryotic structure and provides a molecular basis for understanding the etiology of human ciliopathies.

This study was conducted with IRRF support – Alan Brown, PhD, 2018 and 2019.
Dr. Brown is a member of the faculty of the Department of Biological Chemistry and Molecular Pharmacology at Harvard Medical School in Boston. He holds a B.Sc. in Biochemistry from the University of Warwick, Coventry, UK; and a PhD in Biochemistry from the University of Cambridge, Cambridge, UK